Blog post #2 – First Scientific Entry
November 3rd, 2018
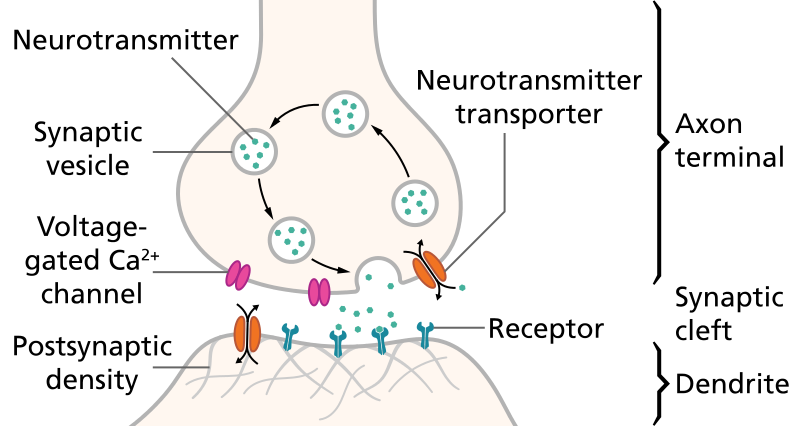
Parkinson’s Disease (PD) is a “a chronic, progressive degenerative neurological disease” that affects over 100,000 Canadians (“Parkinson’s Disease”). In particular, PD involves the incurable and progressive loss of “dopaminergic neurons in the substantia nigra” which regulate the production of dopamine, the chemical responsible for neurotransmissions throughout the body – in the midbrain (K). The function of neurotransmitters, such as dopamine is to regulate motor and non-motor conditions within the human body, such as muscle movement, and cognitive ability. However, the degeneration of these transmitters can make these processes messy, with common symptoms including tremors, muscle rigidity, depression and cognitive impairment (DeMaagd and Philip).
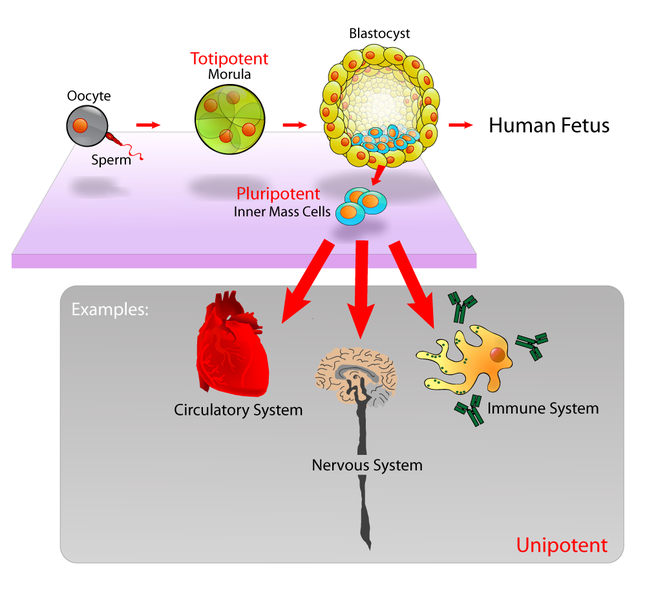
Although there is no known cause or cure for PD, the area of regenerative medicine has recently investigated the use of a few interesting treatment methods using stem cells. One such treatment of PD is the use of embryonic stem cell (ESC) therapy. Stem cells are “cells from which all other cells with specialized functions are generated” including neurons (“Stem Cells”). The human embryo, in its blastocyst stage, is made up almost entirely of these cells in the inner cell mass (ICM), which eventually specialize and divide into a fully functioning human body!
Scientists must first extract these stem cells from a blastocyst-stage embryo, typically donated to labs from fertilization clinics with excess embryos created in vitro, or, within a culture dish, as opposed to within the human body. Once scientists have carefully extracted the stem cells from the embryo, and have placed them within individual culture dishes, they will add specific growth factor proteins like nutrient-rich culture fluids, in a process called three-dimensional culturing (“Stem Cell Differentiation”). In order to create dopamine producing neurons to treat PD, scientists will again transfer these stem cells, which have now formed clusters in their hormone-rich culture dishes called embryoid bodies (EBs) into new dished coated with Matrigel (Cho). From here, the EBs will evolve, or differentiate independently, into purposed nerve cells as indicated by specific neuro markers within the culture dish, assisted by the Matrigel (Cho).
Eventually, surgeons will inject these differentiated stem cells that produce dopamine into patients with PD, via a catheter. While this form of treatment is currently not commercialized, studies are currently underway to test and validate the usage of this method, in order to analyze the risks it may present as a possible medical treatment. A current advantages of using this method of treatment is that these artificial dopaminergic (DA) neurons, created from ESCs, replicate the exact function of natural ones. This means that these neurons have the potential to effectively begin the production of dopamine again in patients with PD. However, current limitations to this method include the risk of immune rejection by the patient (Sun). The genetic makeup of these artificial DA neurons are not an exact match with the patient, therefore, the patient has a risk of rejecting the cells, but also has a risk of developing brain tumours, which can be fatal (Sun). The uncontrolled division of these injected cells can lead to blood clots in the brain, which is a prominent risk holding this treatment method from commercial use.
Overall, there is a lot of work still needing to be done in order to get embryonic stem cell therapy in clinical use around the world. I was inspired to write this blog post after learning about stem cells in my biology class. Genetics and microbiology/immunology have always been my favorite parts of science class, and so I was compelled to take the lens of a scientist to write this blog. My fascination with stem cells drove me to research more about its clinical use, and where scientific research is at in terms of approving it as a valid medical treatment. In the future, I hope to conduct research in the field of cellular and molecular immunology/medicine in order to learn more about how science is evolving, and when the global population can expect to see a breakthrough in commercialized medical treatment. Ibreez Asaria FSL Vancouver Discover Year 2018/2019
Works Cited:
- Cho, Myung Soo, et al. “Highly Efficient and Large-Scale Generation of Functional Dopamine Neurons from Human Embryonic Stem Cells.” The National Academy of Sciences, vol. 105, 28 Feb. 2008, pp. 3392–3397, www.ncbi.nlm.nih.gov/pmc/articles/PMC2265201/pdf/zpq3392.pdf, Accessed 2 Nov. 2018.
- DeMaagd, George, and Ashok Philip. “Parkinson’s Disease and Its Management.” Journal of Pharmacy and Therapeutics, MediMedia USA, Inc., Aug. 2015, www.ncbi.nlm.nih.gov/pmc/articles/PMC4517533/. Accessed 3 Nov. 2018
- K, Ozawa. “Gene Therapy of Parkinson’s Disease Using Adeno-Associated Virus (AAV) Vectors.” Journal of Neural Transmission , 2000, pp. 181–191. National Center for Biotechnology Information. Accessed 3 Nov. 2018
- Sun, Qisi, et al. “The Potential and Challenges of Using Stem Cells for Cardiovascular Repair and Regeneration.” Genes and Diseases, vol. 1, no. 1, 28 July 2014, pp. 113–119., www.sciencedirect.com/science/article/pii/S2352304214000087. Accessed 3 Nov. 2018.
- “Parkinson’s Disease.” UCB, UCB Canada Inc., www.ucb-canada.ca/en/Patients/Conditions/Parkinson-s-Disease. Accessed 3 Nov. 2018.
- “Stem Cell Differentiation.” Curiocity, Let’s Talk Science, www.explorecuriocity.org/Portals/2/Themes/Stem%20Cell%20Differentiation.pdf. Accessed 3 Nov. 2018.
- “Stem Cells: What They Are and What They Do.” Mayo Clinic, Mayo Foundation for Medical Education and Research, 23 Mar. 2013, Accessed 3 Feb. 2018. www.mayoclinic.org/tests-procedures/bone-marrow-transplant/in-depth/stem-cells/art-20048117. Accessed 3 Nov. 2018.
Stem cell therapy provides great hope for patients who are suffering from a wide range of diseases, from cancer to Parkinsons to Alzheimer’s and more. With stem cell therapy, the damage done by these disease can be reversed or even stopped entirely.