Laurence Xie, Life Science
Abstract
Having threaded the earth for a long time, brine shrimp (Artemia) have gone from not only residing in the extremely saline conditions of some saltwater lakes, but have also been put to use as a source of nutrition for the larviculture of fish and shellfish , in lethality assays for plants and pesticides, and even to study embryonic development in space). However, they may now be threatened by changes in the environment such as acid rain. This project aims to test the effects of soaking Artemia cysts in different concentrations of acid rain solutions on their hatch rate afterwards. 24 hours, 48 hours, and 70 hours hatch time results suggest that exposure to acid rain may result in a higher short term hatch rate at the cost of quality and long term survival rate of nauplii.
Introduction
As the aquaculture industry continues to grow larger each day, the demand for nutritious and fast-producing live food to sustain fish larvae has become a necessity to keep up with market demands: Artemia (also known as brine shrimp) nauplii (larvae) have been relied on as a main source of live fish food in aquaculture because of their availability as storable instant live food (Stappen 1996). This can be achievable because of the Artemia cysts, which allow Artemia to be hatched years after they were first laid (Stappen 1996). Not only are the Artemia cysts easily stored for prolonged durations of time but they are also considered storable instant live food because of their ability to hatch roughly 24 hours after being rehydrated in salty water with sufficient conditions (Treece 2000). Furthermore, after the dehydrated cysts are rehydrated, the yolk sack that is still attached to the nauplii contains many essential nutrients including various proteins and calories, with studies showing that the main value of Artemia as aquaculture feed being in their essential fatty acids: high level of highly unsaturated fatty acids (Treece 2000). One potential problem brought by the use of Artemia nauplii on the international market is that, although Artemia cysts production exists pretty much everywhere, a large portion comes from just the Great Salt Lakes (Stappen 1996). This means that the aquaculture industry could potentially take a big hit should climate or environmental issues start to affect the production of Artemia in the Great Salt Lake of Utah. One such environmental issue is acid rain, which is a mixture of sulfuric and nitric acids that can cause big problems amongst aquatic ecosystems (Singh & Agrawal, 2008). This project investigates the changes in hatch rates of dehydrated Artemia cysts being subjected to different concentrations of a simulated acid rain solution (sulfuric acid) to imitate possible impacts of acid rain on Artemia populations in the future.
Materials and Methods
Preparation of Different pH Groups
The different pH solutions were created from a simulated acid rain solution (Diluted H2SO4 with a pH of ~4.56) diluted through a titration consisting of roughly 150 mL of demineralized water (Aquafina) in a 500 mL beaker and a biuret filled up with the acid rain solution. This resulted in the three test groups with pH levels of 4.56 (undiluted acid solution), 5.51, and 6.56. The control group’s solution consisted of only demineralized water (Aquafina).
Preparation of environment for hatching Artemia cysts
The solution used to hatch the Artemia cysts was created using 35 g of aquarium salt dissolved in 1 L of spring water (Pure Life) to make a solution with a salinity of 35 ppt. The saline solution was then placed into a transparent 2.5 L plastic box. This step was repeated 4 times for the 4 different test groups. An air pump was then suctioned to the bottom of the plastic box. A light source was then shined onto the plastic boxes. A temperature of roughly 20 degrees Celsius was also maintained within the environment of the boxes.

Figure 1. Fully prepared hatching set up before the introduction of cysts.
Soaking and Hatching of Artemia cysts
Since there was only a dual air pump, only two groups of Artemia could be hatched at a time. The first 2 groups tested were the Artemia cysts soaked in pH levels of 4.56 and 6.56 (Group A); This was followed by the cysts soaked in a pH of 5.51 and the control (Group B). Following the preparation of the test group solutions, the solutions were placed in their own 250 mL beakers. 1.00 g of Artemia cysts was then added to each beaker and left to soak for 24 hours. After the 24 hour soak period, a funnel with a coffee filter on top was used to separate the cysts from the solutions. The cysts were then given a light rinse using spring water before being placed into the plastic boxes containing the saline solution.

Figure 2. Artemia cysts soaking in solution group 4.56

Figure 3. Artemia hatching set up with presoaked cysts.
Sampling of Artemia nauplii
Prior to the collection of the samples, the air pump was turned off and the water was thoroughly mixed to even out the amount of shrimp in the 1 L of saline solution. Population samples were then taken at 24, 48, and 70 hours into the experiment for every group. One 1 mL sample was taken for every group at 24 and 70 hours so hatching time could be considered, whereas 5 were taken for every group at 48 hours after the initial introduction of the cysts into the hatching boxes as these samples were the main focus of the experiment. The shrimp were then placed into a freezer to stop their movement before the population count.
Results
The experiment was run for 70 hours two different times due to equipment limitations. In the first 70 hours, group A – which consists of tests groups 4.56 and 6.56 – was hatched. In the second set of 70 hours, group B – which consists of test groups 5.51 and control – was hatched.
Table 1. Raw data of sampled Artemia populations 24 hours into experiment
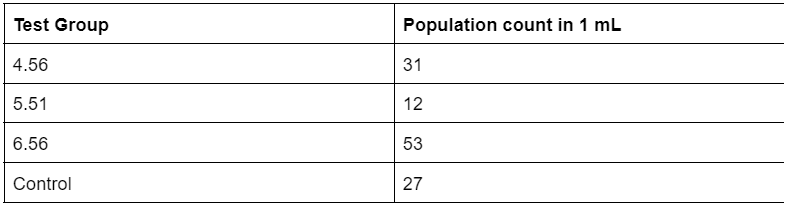
Table 2. Averages of sampled Artemia populations in 1 mL 48 hours into experiment
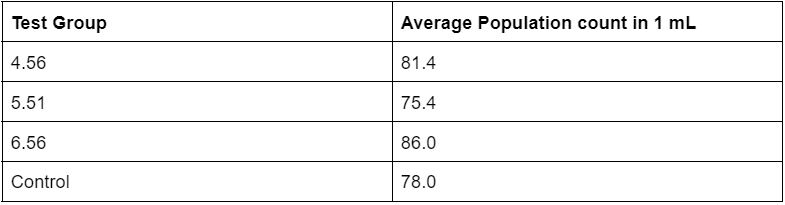
Table 2 consists of an average population count calculated from 5 sets of 1 mL samples. 5 samples were chosen for the 48 hour time frame as it was the main focus in terms of given hatching time.
Table 3. Raw data of sampled Artemia populations 70 hours into experiment
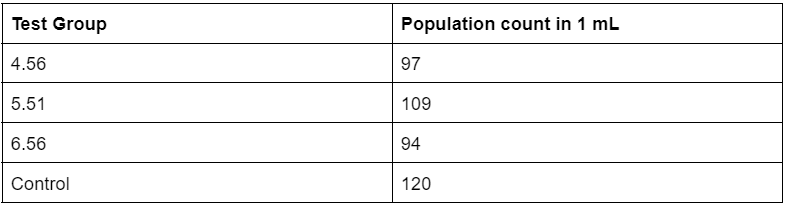


Figure 4. Test groups 4.56 and 6.56 before (top image) and after (bottom image) 24 hour soak
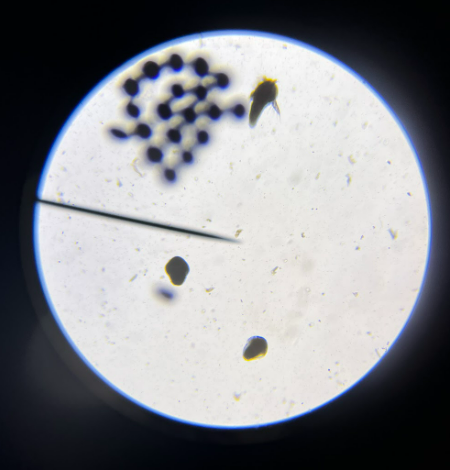
Figure 5. Part of a 48 hour sample of the 5.51 test group containing cyst shells, nauplii, and emerging embryos
Discussion
The overall results of the population sampling at 24 hours showed that the test group with a pH of 6.56 had the highest hatch rate, followed by 4.56, control, and 5.51 (Table 1). These results suggest that, at 24 hours into hatching, the acid rain soak was not completely preventing the hatching of any Artemia groups as test groups with low acidity and comparatively higher acidity all showed signs of hatching in decent quantities. At the 48 hour mark, the population sampling showed a somewhat similar pattern to the sampling taken at 24 hours, with test groups 6.56 and 4.56 holding the highest average population sample in 1 mL. However, qualitative observations of the quality of nauplii and cysts showed that test group 4.56 had noticeably more damaged and fragmented pieces of nauplii. This suggests that the soak in the more acidic solution may have weakened the state of the suspended embryos within the cysts, or caused some other form of damage that ultimately led to a general weakening of the embryos. Finally, at the 70 hour mark, the population sampling results showed that the control group held the highest population count in 1 mL of solution, followed by test group 5.51, 4.56, and 6.56.
In terms of pH and its effects as a factor, the overall results suggest that, in the short term and under the given conditions of this experiment (eg. lower temperature than ideal hatching temperature), the more acidic groups showed higher population counts: The test group with a pH of 6.56 ended with the highest population count after 24 and 48 hours, which could be related to how Artemia cysts exposed to higher concentrations of sulfuric acid for short time periods can be decapsulated and can end up with a higher hatch rate (Hoseini et al. 2013) as it ends up with a higher hatch count than the control group with normal spring water. At the same time, the significant increase in fragmented or damaged nauplii in the 4.56 test group shows that the quality and post-hatch survivability of the Artemia nauplii may be decreased by lower pH levels as Artemia cysts hatch much more favourably in slightly basic conditions (Camargo et al. 2005). The impact of the concentration in the soaking solution can also be observed in Figure 4 as it outlines the change in water quality (from a clear to a brown mixture similar to the colour of the eggs) after the 24 hour soak period, revealing the possibility that parts of the cysts were weakened or fell off as a result of being soaked in an acidic solution. On the other hand, at 70 hours the results completely changed to the opposite of those at 24 and 48 hours with the control (spring water) group having the highest population sample. These observations suggest that short term exposure to acid rain allows for a generally higher hatch rate for Artemia (possibly from decapsulation) but also a lower rate of long–term survival due to the damages done to the nauplii and eggs which prefer to be hatched in a slightly more basic environment.
In terms of effect on the aquaculture industry, the overall results suggest that the soaking of Artemia cysts in acid rain prior to hatching increases the hatching rate, with higher acidities within the test groups providing higher short term hatch rates. However, qualitative observations suggest that a soak in acid rain may not only cause the cysts to be worn down but also cause damage to the embryo such as missing body parts or underdevelopment. These damages could impact their ability to act as feed sources as some may never correctly hatch out of their cysts or reach their maximum state of nutritional value.
In terms of sources of errors, it is likely that the environments of the two different time periods of the experiment ended up having an effect on the hatch rate of the different groups as quite a difference can be spotted between groups A and B, with group A (4.56 and 6.56) possessing the higher numbers in all of the early time periods before being surpassed by group B in Table 3. A better controlled environment with a stable temperature – could be upgraded using an aquarium heater – and a consistent light source would help ensure that the environment remains consistent throughout the experiment. A bigger air pump or more air pumps would also allow all the tests to be run at the same time, which would in turn also make the experiment more controlled. Human error is also likely to have occurred when counting the hatched nauplii as this was done mostly with the naked eye and with quite a lot of nauplii per sample; a smaller sample size along with a magnifying glass could lower the chances of such errors.
Further tests involving longer observation periods, longer soak times, and multiple trials could lead to more consistent and specific parameters that could be used to better understand the effects of the increasing amount of acid rain in the world on the hatch rate of Artemia cysts and methods that can be implemented to protect them.
References
Browne, R. A., & Wanigasekera, G. (2000). Combined effects of salinity and temperature on survival and reproduction of five species of Artemia. Journal of Experimental Marine Biology and Ecology, 244(1), 29–44. https://doi.org/10.1016/s0022-0981(99)00125-2
Burns, D. A., Aherne, J., Gay, D. A., & Lehmann, C. M. B. (2016). Acid rain and its environmental effects: Recent scientific advances. Atmospheric Environment, 146, 1–4. https://doi.org/10.1016/j.atmosenv.2016.10.019
Camargo, W. N., Durán, G. C., Rada, O. C., Hernández, L. C., Linero, J.-C. G., Muelle, I. M., & Sorgeloos, P. (2005). Determination of biological and physicochemical parameters of Artemia franciscana strains in hypersaline environments for aquaculture in the Colombian Caribbean. Saline Systems, 1(1). https://doi.org/10.1186/1746-1448-1-9
Hoseini, S. M., Abolhasani, M. H., & Ghorbani, R. (2013). Sulfuric acid treatment for Artemia cyst decapsulation. NPAJournals.
Krishnaraju, A. V., Rao, T. V. N., Sundararaju, D., Vanisree, M., Tsay, H.-S., & Subbaraju, G. V. (2005). Assessment of Bioactivity of Indian Medicinal Plants Using Brine Shrimp (Artemia salina) Lethality Assay. International Journal of Applied Science and Engineering, 3(2), 125–134. https://doi.org/10.6703/IJASE.2005.3(2).125
Michael, A. S., Thompson, C. G., & Abramovitz, M. (1956). Artemia salina as a Test Organism for Bioassay. Science, 123(3194), 464–464. https://doi.org/10.1126/science.123.3194.464.a
Sarah, Q. S., Anny, F. C., & Misbahuddin, M. (2017). Brine shrimp lethality assay. Bangladesh Journal of Pharmacology, 12(2), 5. https://doi.org/10.3329/bjp.v12i2.32796
Singh, A., & Agrawal, M. (2008). Acid rain and its ecological consequences. Journal of Environmental Biology, 29(1). http://www.jeb.co.in/journal_issues/200801_jan08/paper_02.pdf
Spooner, B. S., DeBell, L., Hawkins, L., Metcalf, J., Guikema, J. A., & Rosowski, J. (1992). Brine Shrimp Development in Space: Ground-Based Data to Shuttle Flight Results. Transactions of the Kansas Academy of Science (1903-), 95(1/2), 87–92. https://doi.org/10.2307/3628023
Stappen, G. V. (1996). 4.1. Introduction, biology and ecology of Artemia. Www.fao.org. https://www.fao.org/3/w3732e/w3732e0m.htm#4.1.%20Introduction
Treece, G. D. (2000, October). Artemia Production for Marine Larval Fish Culture. Artemia Production for Marine Larval Fish Culture; Southern Regional Aquaculture Center. https://www.hatcheryfm.com/documents/20/1325052010_2.pdf
Wu, C. (2014). An important player in brine shrimp lethality bioassay: The solvent. Journal of Advanced Pharmaceutical Technology & Research, 5(1), 57–58. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3960796/#ref2