Millicent Sharman, Life Science
Abstract
In a world brimming with harmful bacteria and diseases lives a potent plant native to China known as Qinghao, or Artemisia annua. Artemisinin, a compound derived from this plant, has saved millions of lives and led to a paradigm shift in antimalarial drug development; artemisinin is also known to be antimicrobial. This experiment was conducted to verify the antimicrobial properties of artemisinin in 90% ethanol, at different concentrations (0.01mg/mL, 0.1mg/mL, 1mg/mL, and 5mg/mL), against E. coli B, a Gram-negative bacterium, and B. subtilis, a Gram-positive bacterium. The results were largely inconsistent due to a variety of random errors, including a presence of cellulose in the capsules used. Based on the success of similar experiments, if this experiment were to be repeated with more standardized methods, the antimicrobial efficacy of artemisinin could be observed more fully.
Introduction
Artemisinin is a natural compound derived from sweet wormwood, or Artemisia annua. It is known to be anti-inflammatory, antifungal, and antitumour, but it is best known for its antimalarial properties, acting as a crucial component of many modern malaria treatments (Kim 2015, Su and Miller 2015). Artemisinin also possesses antimicrobial properties that grant it promise as a natural disinfectant (Appalasamy, et al. 2014).
Some of the most commonly used disinfectants, known as “chemical disinfectants”, have many drawbacks due to their artificial nature, such as irritation and potentially serious damage when in contact with skin, eyes, and the respiratory system (Occupational Safety and Health Council). Furthermore, certain components of disinfectants, such as chlorine, can interact with organic materials in water, forming disinfection by-products, or DBPs (Centers for Disease Control and Prevention). These DBPs may increase risk of cancer, and extremely large amounts of exposure may also lead to liver damage and decreased nervous system activity (Centers for Disease Control and Prevention).
As such, this project was conducted with the goal of substantiating the efficacy of artemisinin as a natural disinfectant. Prior to this project, research into the antimicrobial properties of artemisinin had previously been carried out. In fact, in a 2014 report, artemisinin extracted from Artemisia annua grown in vitro was observed to effectively combat the growth of a variety of Gram-positive and Gram-negative bacteria (Appalasamy, et al. 2014). In a 2015 report, Artemisia annua L. extracts were also observed to contain “antimicrobial substances”, and it was concluded that they “should be considered for use in pharmaceutical products for the treatment of dental diseases” thanks in part to this property (Kim 2015). However, due to the antimalarial capabilities of artemisinin, the majority of research surrounding the compound is primarily related to malaria treatment. Thus, this project was formulated with two aims: firstly, that its results would clarify the viability of artemisinin as a safe alternative to chemical antibacterial treatments, and secondly, that the results would widen the overall amount of data surrounding artemisinin and its antimicrobial properties available in the scientific field. In this experiment, the efficacy of artemisinin in combating growth of both Gram-negative (E. coli B) and Gram positive (B. subtilis) bacteria was tested, through the creation of several artemisinin-ethanol solutions. It was hypothesized that the higher the concentration of artemisinin, the more effective each artemisinin-ethanol solution would be against both E. coli B and B. subtilis.
Materials and Methods
Petri dishes with LB agar were prepared. Four different solutions of artemisinin were prepared from the contents of artemisinin capsules (Provita Nutrition) serially diluted in 90% ethanol, with concentrations as follows: 0.01mg/mL (Solution A), 0.1mg/mL (Solution B), 1mg/mL (Solution C), and 5mg/mL (Solution D). A positive control, pure 90% ethanol (Solution E), and a negative control, distilled water (Solution F), were also collected and set aside. An E. coli B suspension and B. subtilis suspension were made by combining distilled water with a qualitatively consistent amount of bacteria collected on a micropipette tip. Nine petri dishes were prepared with 100μl of the E. coli B suspension, and nine petri dishes were prepared with 100μl of the B. subtilis suspension. Each petri dish was split into six sections, labelled A through F, corresponding to each solution made. Nine sterile antibiotic assay discs were obtained and doused individually on a clean surface in 50μl of Solution A. Each disc was then placed by tweezers in its respective labelled region within a petri dish. This procedure was repeated for the remaining solutions. The discs were stored at room temperature, and zones of inhibition were measured two weeks after initial preparation.
Results
All measurements written below are the diameters of the zones of inhibition surrounding each disc. Each measurement includes the width of the disc itself (6mm). Please see Figure 1 for reference.

Figure 1. E. coli petri dish with drawing
The red line drawn indicates how a zone of inhibition was measured for each disc.
Table 1. Solution Reference Table
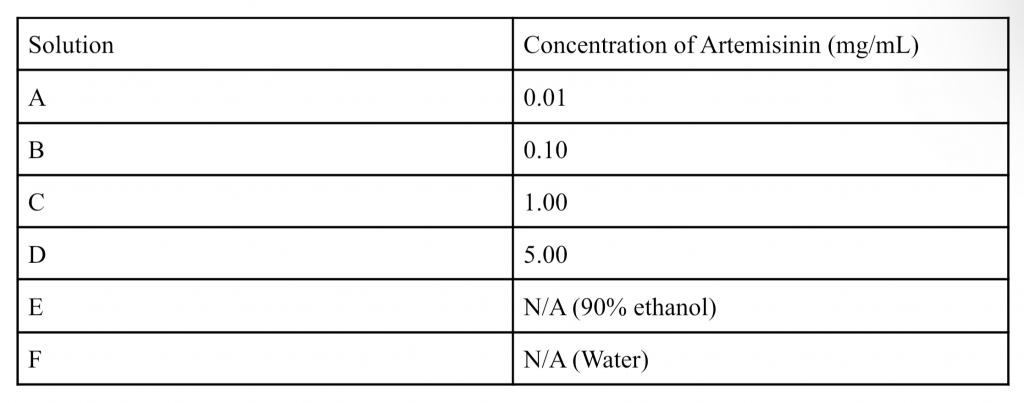

Figure 2. All E. coli B plates
Table 2. Averaged E. coli B data


Figure 3. Graph of Average Zones of Inhibition of E. coli

Figure 4. All B. subtilis plates
Table 3: Averaged B. subtilis data


Figure 5. Graph of Average Zones of Inhibition of B. subtilis
Throughout this experiment, the measured zones of inhibition were highly inconsistent between trials. Because of this, trials in which the positive control within the experiment (Solution E/pure 90% ethanol) failed to create a zone of inhibition wider than the disc itself were excluded from the averaged data seen in tables 2 and 3. This was done because a lack of inhibition outside the disc in the presence of a positive control likely indicates an error within the plate.
Discussion
In Table 2, the average diameter of the zones of inhibition for each solution follows the hypothesized trend, increasing from Solution A to D. As expected, Solution E, the positive control, has a higher average zone of inhibition than Solution F, the negative control. Solution D has a higher average zone of inhibition than Solution E, which may indicate that artemisinin positively affected the inhibition of E. coli B growth with Solution D.
In Table 3, the hypothesized upward trend of inhibition is followed by Solutions B through D. The data for Solution E and F also mirrors the data trend in Table 2. Both Solution C and D also have larger average zones of inhibition than the positive control, once again indicating potential influence from artemisinin. However, Solution A had a larger average zone of inhibition than Solution B, despite having a lower concentration of artemisinin.
The difference between Solution A and B, the inconsistent shapes of each zone of inhibition (visible in figures 2 and 4), and the efficacy of various solutions being lower than that of the positive control, can be attributed to random error. If this experiment were to be conducted again, a more standardized method of applying bacterial solution to each plate and solutions to each disc should be followed. For precision, the McFarland standard should be used in the creation of each bacterial suspension. Additionally, when applying the bacteria to each petri dish, a timer should be used to ensure the bacteria have been evenly and thoroughly spread. Because the application of the solution to the discs occurred on a flat surface, the amount of solution being absorbed by the discs may have varied slightly depending on if the solution stuck to the surface. Thus, if this were to be done again, rather than pipetting each solution onto a disc on a surface, discs should be placed in a solution and soaked for a set amount of time, then directly placed in each petri dish. Finally, the artemisinin should be collected from a different source. In this experiment, solutions were created by dissolving the contents of artemisinin capsules (Provita Nutrition) in ethanol. However, the presence of cellulose in the capsules led to a white precipitate present in each solution. Artemisinin solubility in ethanol-water solutions increases as the concentration of ethanol increases (Wang, et al. 2007). The ethanol used in this experiment was 90% ethanol; while the concentration of ethanol was high, some of the precipitate may still have been artemisinin. Thus, each solution was shaken for ten seconds before being pipetted, to compensate for a potential presence of precipitated artemisinin. However, the amount of artemisinin present on each disc may have consequently varied, and, while unlikely, the zones of inhibition could have been affected by small amounts of cellulose. The averaged results of this experiment partly align with current data in the scientific field. In a 2014 paper on the antimicrobial activity of artemisinin, artemisinin and a precursor bioactive compound were found to “inhibit the growth of [specific Gram-positive and Gram-negative bacteria, including E. coli and B. subtilis] at the minimum inhibition concentration of 0.09mg/mL” (Appalasamy, et al. 2014). This partly aligns with the data in tables 2 and 3; in Table 2, Solution D has a larger average zone of inhibition than Solution E, and in Table 3, both solutions C and D have larger average zones of inhibition than Solution E. This indicates artemisinin may have contributed to the inhibition of bacteria in the specific trials for these solutions. However, solutions A and B (0.01mg of artemisinin per mL and 0.10mg of artemisinin per mL, respectively) had consistently smaller zones of inhibition than Solution E. This does not align with previous research, and is likely a result of random error, as artemisinin has not been previously observed to hinder the efficacy of ethanol as a disinfectant. Unfortunately, due to these varying results between trials, and small sample size, it is impossible to conclusively distinguish between possible random error and the antibacterial effects of artemisinin. However, considering past research, if this experiment were to be repeated with more trials and more standardized methods in place, artemisinin could consistently exhibit statistically significant antibacterial properties.
References
Appalasamy, Suganthi et al. “Antimicrobial activity of artemisinin and precursor derived from in vitro plantlets of Artemisia annua L.” BioMed research international vol. 2014 (2014): 215872. doi:10.1155/2014/215872
“Chemical Disinfectants.” Centers for Disease Control and Prevention, U.S. Department of Health; Human Services, 18 Sept. 2016,https://www.cdc.gov/infectioncontrol/guidelines/disinfection/disinfection-methods/chemical.html#Alcohol.
Chemical Safety in the Workplace: Guidance Notes on Safe Use of Chemical Disinfectants.PDF.https://www.labour.gov.hk/eng/public/os/C/Disinfectants.pdf.
“Disinfection by-Products (DBPs).” Centers for Disease Control and Prevention, U.S. Department of Health; Human Services, 18 July 2022, https://www.cdc.gov/biomonitoring/THM-DBP_FactSheet.html.
Kim, Wan-Su et al. “Anti-inflammatory, Antioxidant and Antimicrobial Effects of Artemisinin Extracts from Artemisia annua L.” The Korean journal of physiology & pharmacology : official journal of the Korean Physiological Society and the Korean Society of Pharmacology vol. 19,1 (2015): 21-7. doi:10.4196/kjpp.2015.19.1.21.
Su, Xin-Zhuan, and Louis H Miller. “The discovery of artemisinin and the Nobel Prize in Physiology or Medicine.” Science China. Life sciences vol. 58,11 (2015): 1175-9. doi:10.1007/s11427-015-4948-7.
Wang, Long-Hu, et al. “Solubility of Artemisinin in Ethanol + Water from (278.2 to 343.2) K.” Chem. Eng. Data 2007, 52, 3, 757–758. doi: 10.1021/je0603426.