Angela Hu, Year 2 Research.
Abstract
The production and disposal of petroleum based plastics poses an abundance of threats to our environment, animals, and human health. Additionally, the consumption of longan (Dimocarpus longan) fruits results in an abundance of longan pits as waste. By extracting its starch to create a biodegradable bioplastic, both of these problems can be addressed; however, there is currently a lack of research concerning the matter. In this experiment, longan bioplastics of various concentrations of glycerol and zinc oxide were tested and compared with a typical cornstarch bioplastic on three criteria: biodegradability, tensile strength, and water absorption. It was found that higher concentrations of glycerol and ZnO seemingly resulted in higher tensile strengths, albeit below that made of cornstarch. No clear trends were noticed in biodegradability or water absorption and more testing will be needed to determine if longan starch bioplastics can serve as an alternative to other plastics.
Introduction
Petroleum based plastics are commonly used for a variety of purposes throughout the world, yet are non-renewable and destructive to the environment (Barney and Dyke 2020). For example, the Great Pacific Garbage patch has reached a size three times that of France (Liu 2018), and Kamilo Beach is covered in 10ft-high high mountains of trash (Barney and Dyke 2020). Plastic debris can harm wildlife, creating choking hazards and remaining indigestible in animal stomachs – globally, around 100 thousand marine animals alone are killed by plastic waste (WWF 2018). Finding an affordable yet durable biodegradable plastic would largely mitigate the issue of an increasing amount of plastic pollution.
A common alternative scientists turn to is starch-based bioplastics, such as those made with the starch from potato peels (Goswami et al. 2015) or a combination of corn starch and banana peels (Sultan and Johari 2017). Starch is highly degradable, renewable, and low cost, but is growing in market demand to account for the increase in world population (Kringel et al. 2019). Fruit wastes can often contain significant amounts of starch that is currently unexplored (Kringel et al. 2019). Longan fruit (Dimocarpus longan) are widely consumed in subtropical areas including China, Thailand, and Malaysia, and belong to the Sapindaceae family (Hu et al. 2018). Typically, large amounts of longan kernels are thrown away as waste, but some research has found that these pits contain high amounts of starch (49%) (Hu et al. 2018). However, there exists a gap in the literature regarding longan starches with respect to bioplastics. Being able to utilize this starch for bioplastics would minimize waste, add profit for companies, and help in meeting the demand for starch (Kringel et al. 2019).
For this project, I explored the feasibility of using longan kernel starches to create bioplastics using various concentrations of glycerol and zinc oxide. To determine if the bioplastic is efficient enough to be widely used as a substitute for existing plastics, biodegradability, tensile strength, and water absorption (to ensure water resistance, such as for when plastics are used to store liquids) were explored. The samples were also compared to a common corn starch bioplastic. It was hypothesized that if the sample contained higher concentrations of glycerol and zinc oxide, then it would have greater tensile strength but the same water resistance and level of biodegradability.
Materials and Methods
Extracting longan starch
Longan pit starch was isolated from longan fruit purchased from T & T (freshfruit). Seeds were washed to remove traces of any adhering pulp and cut in half, resulting in small pieces of approximately 0.75 cm2. These were steeped for 4 hours in distilled water containing citric acid (Amson Naturals) at a concentration of 0.3% w/w, which has been found to produce higher yield and amylose content, typically connected to greater tensile strength, than sodium hydroxide (Jaiswal and Kumar 2015). Steep water was then drained off, and the seeds were ground in a blender with equal parts seeds to water by mass (150 g of each.) The resulting homogenate (figure 1) was passed through a cheesecloth (Olicity) into a bucket and left to settle for 48 hours. The settled starch layer at the bottom of the bucket was scraped off, collected, and then allowed to air dry.

Figure 1: Longan pit Homogenate.
Making longan plastic
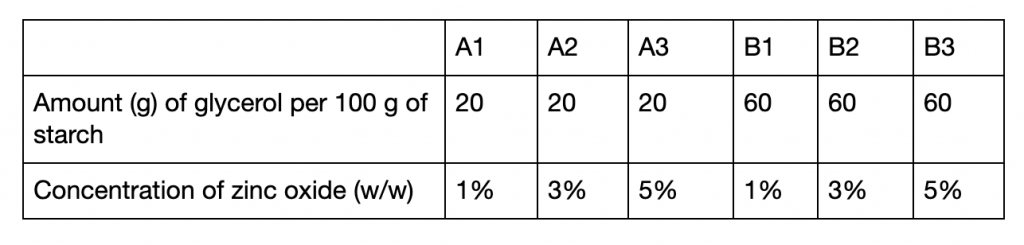
The starch was mixed with glycerol (NOW) as a plasticizer at concentrations of 20 g and 60 g per 100 g of starch, and zinc oxide (Amson Naturals) as an amplifier at 1%, 3%, and 5% w/w (Agustin and Padmawijaya 2017) (figure 2). This mixture was added to distilled water at a starch to water ratio of 1:2 by mass, stirred and heated above a contained boiling bath for 10 minutes, and then spread on petri dishes to dry. Bioplastics with 20 g of glycerol per 100 g of starch were labelled under group A, while those with 60 g per 100 g were in group B. Samples were labelled as 1, 2, or 3 depending on if they contained 1%, 3%, or 5% of zinc oxide (w/w) respectively (table 1).

Figure 2: Batch one of the longan plastic
Table 1: Summary of concentrations of compounds in each bioplastic sample.
Making corn starch bioplastic
To make the cornstarch bioplastic, 27 grams of cornstarch, 150 mL of water, and 15 drops of glycerin were mixed and heated in a microwave at low heat until it began to froth. The mixture was stirred after heating until a sticky precipitate was formed, poured into molds, and left to dry.
Biodegradability test
Each bioplastic was cut into 1 cm x 2 cm size. Biodegradability of the plastics was measured by weighing each sample and burying them in soil at the same depth with the same conditions of pH, temperature, humidity, etc. Each week, for four weeks, the samples were dug up, washed with water, left to dry, and then weighed, before being reburied.
Degradation was calculated with the following formula:
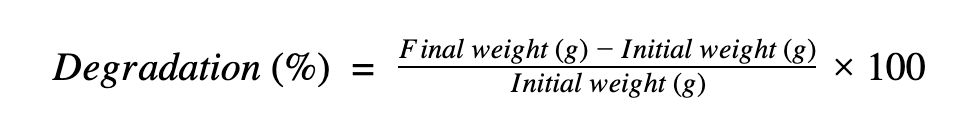
Tensile strength test
Each sample was firmly clamped between two clamps (Neewer) and suspended. A spring balance capable of measuring up to 5 N was hooked over the plastic and slowly pulled downwards until the plastic broke. A slow motion camera was used to more accurately determine the force required to tear the plastic. Three trials of each plastic were conducted, and the data was averaged. Tensile strength was calculated by the force (N) divided by the thickness of the plastic (mm).
Water absorption test
The initial weight of each sample was recorded. The samples were then placed into containers containing water at room temperature. After 24 hours, the samples were taken out from the water, dried, and weighed.
The amount of water absorption was calculated as a percentage change in
weight from the initial weight:
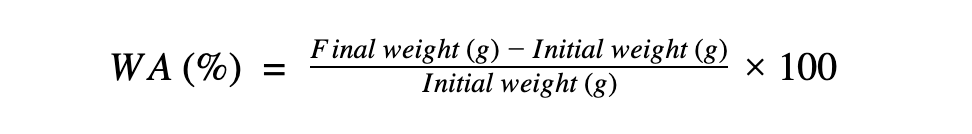
Results
Biodegradability test
The degradation of each bioplastic sample over time was measured.
Table 2: Measurements of the mass of 7 samples across 4 weeks as a test of
biodegradability
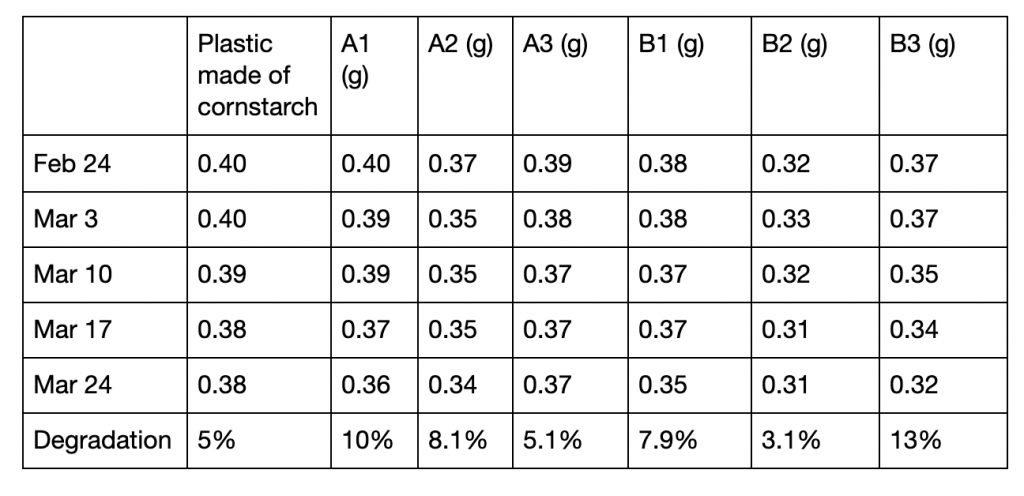
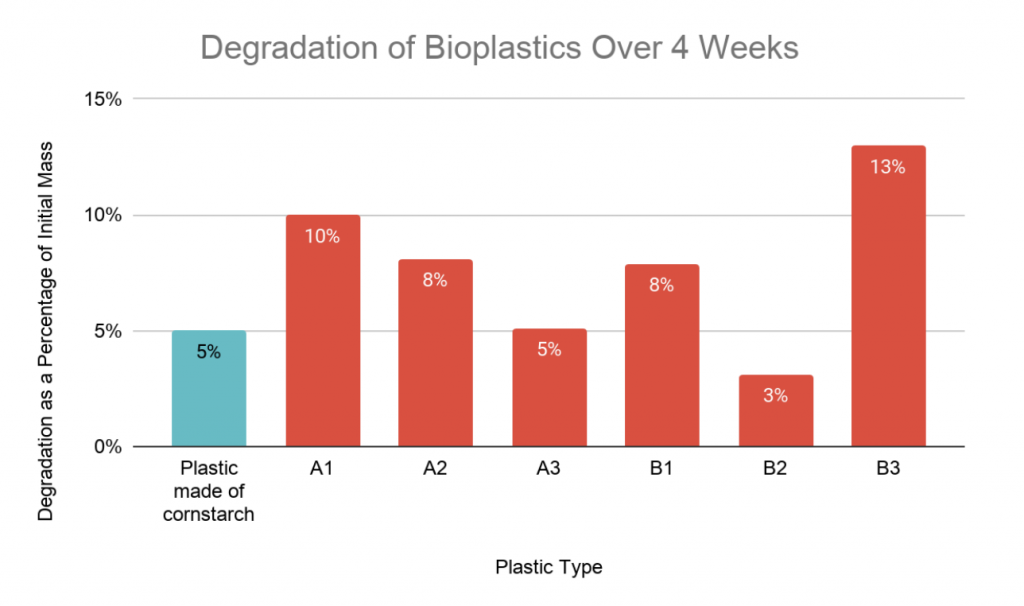
Figure 3: Bar graph representation of the degradation of each bioplastic sample.
Besides sample B2, all longan starch bioplastics resulted in higher rates of degradation than that made of cornstarch (table 2). In bioplastics in group A, a higher concentration of zinc oxide correlated with a lower rate of biodegradation. No clear trend can be seen between B1, B2, and B3 (figure 3).
Tensile strength test
The tensile strength of each sample was measured.
Table 3: Tensile strength test of bioplastics
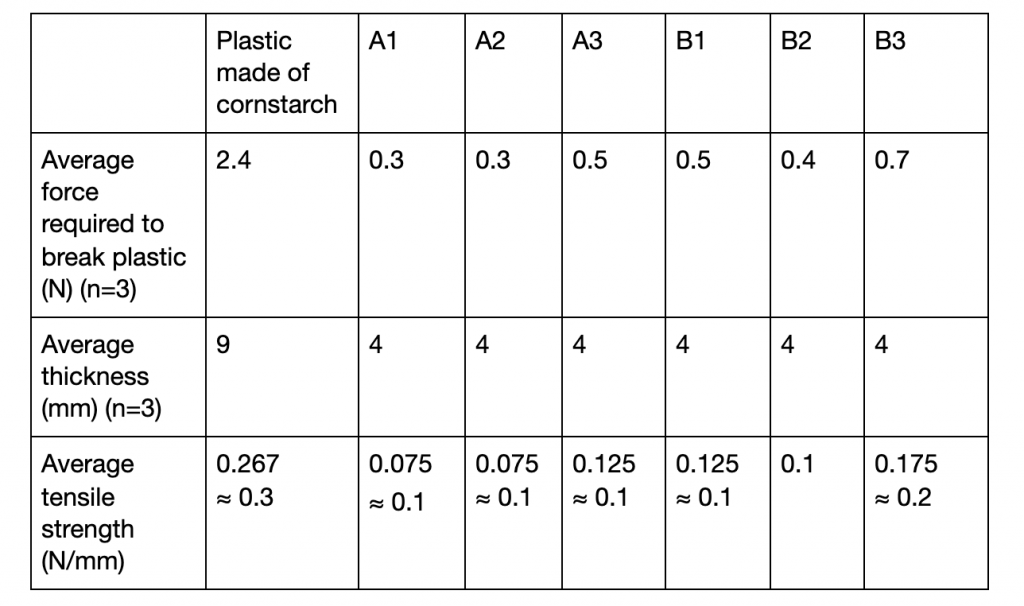
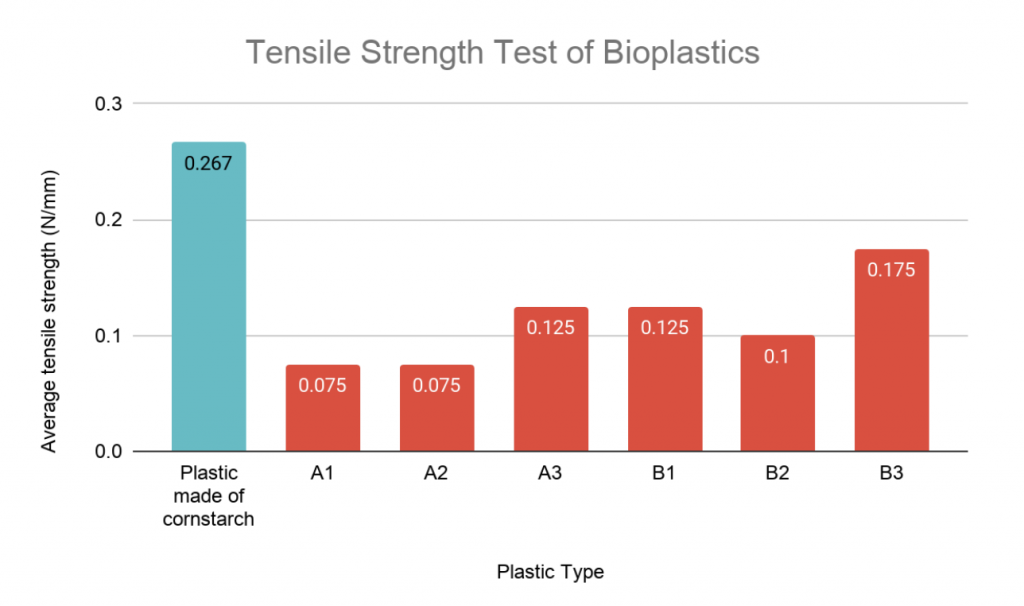
Figure 4: Bar graph representation of the average tensile strengths of each bioplastic sample.
All longan bioplastics had a tensile strength (rounded to one significant figure) of 0.1 N/mm except that of sample B3 (table 3). The bioplastic made of cornstarch had the greatest average tensile strength at nearly 0.3 N/mm (figure 4).
Water absorption test
Water absorption over 24 hours was measured for each sample.
Table 4: Water absorption test of 7 samples measured over the course of 24 hours. 0g and negative water absorption percentages indicate the plastic fell apart.

All samples that resulted in usable data had a higher than 50% water absorption rate (table 4). Samples A1 and A3 demonstrated a water absorption percentage higher than 100%, and samples A2 and B3 fell apart into more than two pieces in the water. Longan samples A1, A3, and B1 have higher absorption percentages than that of the cornstarch bioplastic.
Discussion
Results show that nearly all longan bioplastics are more biodegradable than cornstarch bioplastics, and the bioplastic made of the highest concentrations of both glycerol and zinc oxide (sample B3) resulted in the highest rate of degradation. However, the sample with the lowest concentration of both demonstrated the second highest level of biodegradability, leading to no noticeable trend. Despite this, all samples are promising as being an environmentally friendly alternative to conventional plastics. Only one trial was conducted in order to allow for higher starting masses of each sample; this was done so that the change in mass would be more noticeable. In future experiments, more measurements should be taken over a longer period of time before making conclusions about precise biodegradability levels.
The test should also be contained in a more controlled environment to avoid the impact of fluctuations in light, humidity, and temperature on biodegradability, something that was difficult to do due to the experiment being conducted at home.
The bioplastic made of cornstarch displayed the highest tensile strength, while all longan bioplastics displayed a tensile strength of 0.1 N/mm or 0.2 N/mm. Of the longan bioplastics, those with the highest concentration of ZnO at each amount of glycerol demonstrated the highest tensile strength. This is supported by Abdullah et al. (2020), who concludes that zinc oxide acts as a filler to strengthen starch-based bioplastics. However, sample B2 seemingly broke this trend, which could be a result of needing more precise data points. Test method ASTM can be applied, a common tensile strength test. Bioplastics in group B with higher levels of glycerol also had, on average, higher strengths, which aligns with research (Goswami et al. 2015) that glycerol (propane-1, 2, 3-triol) provides flexibility, acting as a plasticizer and disconnecting polymer chains from one another.
All bioplastics demonstrated fairly high water absorption percentages, with only sample B1 having one below that of the cornstarch bioplastic. This may be attributed to the hydrophilic nature of biopolymers (Sultan and Johari, 2017) and glycerol. Notably, two samples fell apart in the water. This resulted in inconclusive data, and further testing should be conducted to determine if this is an outlier. This may be due to traces of longan pit pulp still remaining in the extracted starch, disintegrating in the water and weakening chemical bonds.
For future experiments, a centrifuge should be used to guarantee extracted longan starch is not contaminated with leftover pulp. From the water absorption data available, sample B2 is the most viable for commercial use, due to higher water resistance. Taken together, further testing is needed to better characterize the effect of different concentrations of glycerol and zinc oxide on biodegradability, tensile strength and water resistance on longan pit starch bioplastics. However, the ability for all samples to biodegrade seems promising. Bioplastics with higher concentrations of both compounds appear to exhibit higher tensile strengths, which is consistent with the initial hypothesis. This experiment is a first step to fill the data gap regarding longan pit starches with respect to commercially viable substitutes for petroleum based plastics.
In particular, improving the tensile strengths of these bioplastics will need to be explored before determining if they can be a suitable replacement for any of the plastics currently in the market. To ensure accuracy, future research involving more advanced laboratory equipment can conduct trials on a larger scale, such as over extended periods of time or with different concentrations of compounds.
References
Abdullah, Akbar Hanif Dawam, et al. “Harnessing the Excellent Mechanical, Barrier and Antimicrobial Properties of Zinc Oxide (Zno) to Improve the Performance of Starch-Based Bioplastic.” Polymer-Plastics Technology and Materials, vol. 59, no. 12, Aug. 2020, pp. 1259–67. Taylor and Francis+NEJM,
doi:10.1080/25740881.2020.1738466.
Agustin, Y. E., and K. S. Padmawijaya. “Effect of Glycerol and Zinc Oxide Addition on Antibacterial Activity of Biodegradable Bioplastics from Chitosan-Kepok Banana Peel Starch.” IOP Conference Series: Materials Science and Engineering, vol. 223, July 2017, p. 012046. DOI.org (Crossref),
doi:10.1088/1757-899X/223/1/012046.
Barney, Liz, and Michelle Broder Van Dyke. “Welcome to Hawaii’s ‘Plastic Beach’, One of the World’s Dirtiest Places.” The Guardian, Guardian News and Media, 10 Jan. 2020, http://www.theguardian.com/us-news/2020/jan/10/kamilo-beach-plastic-hawaii-pollution. Accessed 7 May 2021.
Goswami, Garima, et al. “Bioplastics from Organic Waste.” International Journal of Engineering Research & Technology, vol. 3, no. 23, Apr. 2018.
Hu, Ziman, et al. “Hierarchical Structure, Gelatinization, and Digestion
Characteristics of Starch from Longan (Dimocarpus Longan Lour.) Seeds.”
Molecules, vol. 23, no. 12, Dec. 2018. PubMed Central,
doi:10.3390/molecules23123262.
Jaiswal, P., and K. Jayaram Kumar. “Physicochemical Properties and Release
Characteristics of Starches from Seeds of Indian Shahi Litchi.” International
Journal of Biological Macromolecules, vol. 79, Aug. 2015, pp. 256–61.
ScienceDirect, doi:10.1016/j.ijbiomac.2015.04.050.
Kringel, Dianini Hüttner, et al. “Fruit Wastes as Promising Sources of Starch: Extraction, Properties, and Applications.” Starch – Stärke, vol. 72, no. 3–4, 2020, p. 1900200. Wiley Online Library,
doi:https://doi.org/10.1002/star.201900200.
Liu, Marian. “Great Pacific Garbage Patch Now Three Times the Size of France.” CNN, Cable News Network, 23 Mar. 2018,
https://www.cnn.com/2018/03/23/world/plastic-great-pacific-garbage-patch-i
ntl/index.html. Accessed 7 May 2021.
“Plastic in Our Oceans Is Killing Marine Mammals.” WWF, 11 Oct. 2018,
https://www.wwf.org.au/news/blogs/plastic-in-our-oceans-is-killing-marine-mammals. Accessed 7 May 2021.
Sultan, Noor Fatimah Kader, and Wan Lutfi Wan Johari. “The Development of Banana Peel/Corn Starch Bioplastic Film: A Preliminary Study.” Bioremediation Science and Technology Research, vol. 5, no. 1, July 2017, pp. 12–17.